

Pharmaceutical Industries
Product Details:
Product Description
Titanium Dioxide, Why in Pharmaceutical Industries !
-
It occupies the most Vital Roleand plays an essential Role, from Colouring to Packaging, at all stages of a Pharmaceutical product's Lifecycle.
-
It meets the most stringent of requirements governing thesafety of Medicines, including those set by IP, BP, EP, JP & also USP.
-
It performs a number of Important Functionsthat apply to all stages of a Pharma product's lifecycle,
   To making a Medicine easier to identify or take.Â
- It serves the following functions,
As a Pigment :Â It is used to add whitenessor accentuate the boldness of other colours. It helps tablets stand out forboth Medical Professionals and Patients, to differentiate the strengths of thesame Medication.
Coatings : It is an essentialcomponent of the Tablet Coating, necessary to preserve the Safety, Efficacy andQuality of the Active Pharmaceutical Ingredient (APIs), and to provideShelf-Life Stability. It offers protection for Photosensitive Ingredients,which could be damaged by visible light, and also offer protection to the APIs/ Ingredients that may be vulnerable to Ultraviolet (UV) light degradation.
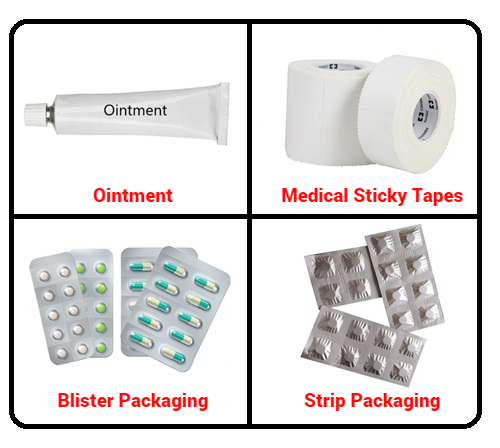
Packaging : It is widely used inprotective films for Tablet Strips and Blisters, as well as in externalCartons. This is because of its ability to scatter light and absorb UVrays, means it is routinely incorporated in the packagings of Medicines tomaintain Shelf Life and prevent any Premature Degradation from Moisture, Heator Light.Â
Applications :Â
It is used in almost all Pharmaceutical Formulation because of itsfollowing characteristics ;
- It adds Whiteness to Tablets & Capsules.
- It is used to accentuate the Boldness of other Colours.
- To differentiate the different strengths of the same Medicines.
- In the coating it is used to preserve the Safety, Efficacy & Quality ofthe APIs.
- It also provides Shelf Life Stability.
- It offers Protection for Photosensitive Ingredients.
- It is used in packagings of Medicines tomaintain Shelf Life and prevent any Premature Degradation from Moisture, Heat or Light.
Ultra Pure Product, Heavy Metals Contents, BLQ
[Tested by Independent ProfessionalTesting Laboratory, NABL & FSSAI approved  (By one Lab.)]
|
Sr. No. |
Parameters |
 Units |
Results of Analysis |
Limits as per FCC |
|
1 |
Lead |
mg/kg |
BLQ |
Max. 10 |
|
2 |
Cadmium |
mg/kg |
BLQ |
Not Specified |
|
3 |
Copper |
mg/kg |
BLQ |
Not Specified |
|
4 |
Arsenic |
mg/kg |
BLQ |
Max. 1 |
|
5 |
Tin |
mg/kg |
BLQ |
Not Specified |
|
6 |
Methyl Mercury as Mercury  |
mg/kg |
BLQ |
Not Specified |
|
7 |
Mercury |
mg/kg |
BLQ |
Max. 1 |
BLQ - Below Limit of Quantification
LOQ (Limit of Quantification) for Heavy Metals =0.5mg/kg
FCC : Food Chemical Codex
Heavy Metals Contents & Other CriticalParameters, Tested by Independent Professional Testing Laboratory, NABL &FSSAI approved  (By Theother Lab.)
|
Sr. No. |
Test Parameters |
 Test Results |
Specification |
|
1 |
Loss on Drying |
0.31% |
NMT 0.5% |
|
2 |
Loss on Ignition |
0.26% |
NMT 1.0% |
|
3 |
Water Soluble Substance |
0.17% |
NMT 0.5% |
|
4 |
Acid Soluble Substance |
0.31% |
NMT 0.5% |
|
5 |
Lead |
Not Detected |
NMT 10.0 mg/kg |
|
6 |
Cadmium  |
Not Detected |
NMT 1.0 mg/kg |
|
7 |
Mercury |
Not Detected |
NMT 1.0 mg/kg |
|
8 |
Arsenic |
Not Detected |
NMT 1.0 mg/kg |







